VIDEO: http://www.youtube.com/watch?v=onCvpFqNhqg&feature=youtu.be TRANSHUMANISM
US company to offer methods for the selection blue eyes, longer life and athletic properties for offspring
The US company 23andMe has received a US patent for the selection of human sperm cells and oocytes (US8543339). Genetic data can be collected from which potential parents can choose donors according to criteria such as eye colour, longer life span and athletic properties.
“The genetic identity of humans never should be selected according to fashion, trends or even commercial interests. Eugenic selection governed by commercial interests is in fundamental conflict with human dignity”, Christoph Then says for Testbiotech. “Any business based on creation of designer babies should not be fostered by patent law.”
The patent was also filed in Europe and other regions (WO 2010/065139). However, it is not likely to be granted in Europe because there is a prohibition of patents that are against morality and public order. Further the patent can be classified as a business method which is - contrary to the US – regarded as not patentable in Europe. Meanwhile the application seems to have been withdrawn in Europe.
Nevertheless, the European Patent Office, EPO, has already granted several patents, which are problematic from this ethical point of view. For example, Ovasort received a patent in 2011 (EP1263521) on the selection of human sperm cells. In 2009, Merck Serono received a patent on the usage of human oocytes (EP1794287). In both cases Testbiotech has filed oppositions – however the EPO has not made any decisions yet.
Christoph Then, 004915154638040, info@testbiotech.org
NEWS ON CLONING AND STEM CELL RESEARCH
World's Second Cloned Baby Is Born, Raelians Say
Updated 11:40 AM ET January 4, 2003
By Eric Onstad
AMSTERDAM (Reuters) - The world's second cloned baby was born on Friday to a Dutch woman, the head of the Raelian sect in the Netherlands said on Saturday.
"A baby girl was born yesterday evening. The baby is healthy and the mother, too," Bart Overvliet told Reuters by telephone.
The woman, a lesbian, is in the Netherlands with her partner, but the birth might have taken place in another country, he added.
He said the child was created by Clonaid, the same cloning firm that claimed last month to have organized the birth of the first human clone, named "Eve," to a 31-year-old U.S. woman.
Clonaid's initial claim sparked widespread skepticism among scientific experts, and the company has yet to provide DNA samples or other evidence to support its assertions about last month's birth.
Clonaid was established by the Raelian movement, a religious group that believes aliens landed on Earth 25,000 years ago and started the human race through cloning.
The founder of the movement, Claude Vorilhon, who calls himself "Rael," told CNN on Friday that Clonaid and the Raelian movement were "very different" and he could not personally vouch for the accuracy of Clonaid's claims.
Overvliet said the Dutch woman involved in the latest birth plans to raise the baby with her partner and is not a member of the Raelian movement.
"It's a lesbian couple, but she is not a member of the religion, she got in contact with Clonaid by herself," said Overvliet, a 45-year-old Amsterdam salesman.
Cloning a human is forbidden in the Netherlands, but nothing in the law forbids the birth of a cloned baby, a spokesman for the Dutch Health Ministry said.
FEARS OF DEFECTS
Clonaid, which says it has a list of 2,000 people willing to pay $200,000 to have themselves or a loved one cloned, announced its initial breakthrough on December 27 and said four more cloned babies would be born by the end of January.
Cattle, mice, sheep and other animals have been cloned with mixed success. Some of these animals have shown defects later in life and critics of human cloning say it is unethical to subject a baby to these dangers.
The Raelians dismiss fears about cloned babies suffering health problems as propaganda aimed at impeding the progress of cloning.
"These scientists don't want to let cloning progress, they want to stop it because they are afraid of human cloning. They say on purpose that it has a lot of faults and genetic defects," Overvliet said.
He said Clonaid's work was a logical progression of in-vitro fertilization (IVF), the technique used to help infertile couples have children.
"Human cloning is more of an extension of IVF, cloning of humans is actually less complicated than of animals," he said.
The Raelian Movement, which claims 55,000 followers around the world, has around 30 members in the Netherlands, but none of them so far have expressed interest in being cloned, he said.
Aliens who created humans and then departed for their own planet have been monitoring mankind's progress, Overvliet said.
"They now think we are far enough along in science so we can understand how we were created," he said.
On Thursday, Clonaid chief executive Brigitte Boisselier said in television interviews that DNA tests on the baby born to an American woman had been put off because the parents were anxious about keeping their identity secret.
PARIS (Jan. 3, 2003) - The parents of a newborn claimed to be the world's first cloned human are balking on whether to allow DNA testing on the child, said the head of the cloning company that says it brought the baby to life.
Many experts have expressed skepticism about the company's claim that the baby was a clone, saying they needed to see a DNA matching as proof.
But ``the parents told me that they needed 48 hours to decide yes or no - if they would do it,'' Brigitte Boisselier told French television station France-2 in an interview Thursday.
Boisselier is chief executive of Clonaid, which is linked to a religious sect that believes space aliens created life on Earth. She is also a member of the sect, called the Raelians.
Clonaid has refused to identify the parents or offer any proof that the child - nicknamed ``Eve'' - is a clone. But the company had promised DNA test results to confirm their claim by around the end of this week.
Attempts by The Associated Press to reach Michael Guillen, the freelance journalist who was arranging the DNA testing, were not successful Thursday. A spokeswoman for Clonaid said she was not familiar with Boisselier's broadcast remarks and was unable to comment Thursday.
Boisselier told France-2 the parents were reconsidering whether to submit to testing because of legal action taken in Florida that could result in the cloned child being taken away from them.
Earlier this week, a court in Florida was asked to turn the baby over to state care if it found the baby's health was in danger. Though Clonaid has kept secret the baby's whereabouts, the company held its news conference to announce the clone's birth in Florida, which could give the court jurisdiction, argued lawyer Bernard F. Siegel.
``That is a lot of turbulence for the parents (who) have gone home and just want to have some peace and spend time with their children,'' said Boisselier.
Meanwhile, a second cloned baby was expected to be born somewhere in Europe before Sunday, Boisselier said. She declined to name the country.
Boisselier had previously said that three additional couples were expected to give birth to Clonaid-created clones by early February.
Clonaid, which declines to reveal where its facilities are, was founded in the Bahamas in 1997 by the man who founded the Raelian religious sect. The man, Rael, says he learned about the origin of life on Earth from a visitor from outer space. He says he views cloning as a step toward reaching eternal life.
Clonaid retains philosophical but not economic ties to the Raelians, the company says.
01/02/03 21:26 EST
Copyright 2002 The Associated Press.
HOLLYWOOD, Fla. (Dec. 27, 2002) - A chemist connected to a group that believes life on Earth was created by extraterrestrials claimed Friday to have produced the world's first human clone, a baby girl named Eve.
The 7-pound baby was born Thursday, said Brigitte Boisselier, head of Clonaid, the company that claimed success in the project. She wouldn't say where the baby was born.
Even before the announcement, other scientists expressed doubt that her group could clone a human.
Boisselier, who spoke at a news conference, said the baby is a clone of the 30-year-old American woman who donated the DNA for the cloning process, had the resulting embryo implanted and then gestated the baby. If confirmed, that would make the child an exact genetic duplicate of her mother.
''It is very important to remember that we are talking about a baby,'' she said. ''The baby is very healthy. She is fine, she doing fine. The parents are happy. I hope that you remember them when you talk about this baby, not like a monster, like some results of something that is disugusting.''
Boisselier did not immediately present DNA evidence showing a genetic match between mother and daughter, however. That omission leaves her claim scientifically unsupported.
The group expects four more babies to be born in the next few weeks, another from North America, one from Europe and two from Asia.
She said the baby will go home in three days, and an independent expert will take DNA samples from the baby to prove she had been cloned. Those test results are expected within a week after the testing.
Most scientists, already skeptical of Boisellier's ability to produce a human clone, will probably demand to know exactly how the DNA testing was done before they believe the announcement.
Clonaid was founded in the Bahamas in 1997 by Claude Vorilhon, a former French journalist and leader of a group called the Raelians. Vorilhon and his followers claim aliens visiting him in the 1970s revealed they had created all life on Earth through genetic engineering.
Cloning produces a new individual using only one person's DNA. The process is technically difficult but conceptually simple. Scientists remove the genetic material from an unfertilized egg, then introduce new DNA from a cell of the animal to be cloned. Under the proper conditions, the egg begins dividing into new cells according to the instructions in the introduced DNA.
Boisselier, who claims two chemistry degrees and previously was marketing director for a chemical company in France, identifies herself as a Raelian ''bishop'' and said Clonaid retains philosophical but not economic links to the Raelians. She is not a specialist in reproductive medicine.
Human cloning for reproductive purposes is banned in several countries. There is no specific law against it in the United States, but the Food and Drug Administration contends it must approve any human experiments in this country. Boisselier would not say where Clonaid has been carrying out its experiments. Bush administration officials said in Washington on Thursday they were aware of rumors of an announcement but had no plans to comment on the matter until after the details were known.
In Rome, fertility doctor Severino Antinori, who said weeks ago he had engineered a cloned baby boy who would be born in January, dismissed Clonaid's claims and said the group has no scientific credibility.
The news ''makes me laugh and at the same time disconcerts me, because it creates confusion between those who make serious scientific research'' and those who don't, Antinori said.
''We keep up our scientific work, without making announcements,'' he added. ''I don't take part in this ... race.''
So far scientists have succeeded in cloning sheep, mice, cows, pigs, goats and cats. Last year, scientists in Massachusetts produced cloned human embryos with the intention of using them as a source of stem cells, but the cloned embryos never grew bigger than six cells.
Many scientists oppose cloning to produce humans, saying it's too risky because of abnormalities seen in cloned animals.
AP-NY-12-27-02 0942EST
Copyright 2002 The Associated Press.
Cloned monkey embryos are a "gallery of horrors"
A high percentage of cloned monkey embryos that look healthy are really a "gallery of horrors" deep within, says a researcher at Advanced Cell Technology, the company that last month published the first paper on cloned human embryos.
December 12 , 2001
Sylvia Pagan Westphal, Boston
This could mean that there is something unique about primate eggs that will make cloning monkeys or people far more difficult than cloning other animals. At the very least, the experiments show that there's a lot to learn before primates can be cloned.
Tanja Dominko, who presented the results last week at a conference in Washington DC, did the work before joining ACT, while she was working for the reproductive biologist Gerald Schatten at the Oregon Regional Primate Research Center in Beaverton.
Several groups have been trying for years to clone monkeys, but while the embryos look normal, no one has ever got them to develop further.
Uneven scatter
To try and figure out what was going wrong, Dominko looked at 265 cloned rhesus macaque embryos created by nuclear transfer - plucking out an egg's nucleus and then adding a nucleus from a donor cell. She followed development of the embryos through several divisions, from the two-cell stage until the 32-cell stage.
Though they appeared superficially healthy, the cells in the vast majority of Dominko's embryos did not form distinct nuclei containing all the chromosomes. Instead, the chromosomes were scattered unevenly throughout the cells.
"The surprising thing is that these cells keep dividing," says Dominko. Some embryos developed to the stage known as a blastocyst, but by day six or seven they had started to look abnormal.
The cloned human embryos created by ACT didn't even get this far. Only one reached the six-cell stage.
Trauma of removal
Dominko says that the trauma of removing the nucleus from the egg might be what triggers the defects. Eggs whose nuclei are removed and then put back inside show the same abnormalities, as well as evidence of programmed cell suicide. "This is not to say that normal embryos can't be made, but not on a regular basis," says Dominko.
Ian Wilmut, who cloned Dolly the sheep, told the conference that Dominko's results were not surprising in the light of experience of nuclear transfer in mice and cows. Even in these animals the success rates are not high, so the phenomena observed by Dominko probably occur in them as well - it's just that everyone focuses on the few successes, he says.
Even so, researchers hoping to publish work on nuclear transfer in humans may now have to come up with better evidence that embryos are healthy. William Haseltine, editor of the journal in which ACT published details of its cloned human embryos, now agrees that pictures alone aren't enough.
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
Date: 8/1/2001
US bans human cloning
WASHINGTON - The US House of Representatives has approved a sweeping ban on human cloning, a prohibition that would make it a federal crime to clone people to produce children or to create embryos for medical research.
Saying that human cloning posed too many safety risks and ethical dilemmas, lawmakers in the Republican-controlled House passed the legislation by a 265-162 vote.
The House defeated 249-178 an alternative backed by medical groups and the biotechnology industry that would have permitted human cloning for a type of stem cell research.
The bill, favoured by President Bush, now awaits action by the Democratic-led Senate.
"The moral issues posed by human cloning are profound and have implications for today and for future generations," Bush said in a statement issued by the White House.
"Today's overwhelming and bipartisan House action to prohibit human cloning is a strong ethical statement, which I commend. We must advance the promise and cause of science but do so in a way that honours and respects life," Bush said.
Two groups of scientists have announced plans to clone people to provide children for infertile couples by using the same techniques that gave the world Dolly the sheep in 1997. Lawmakers agreed that human cloning for reproduction should be illegal but battled over whether to permit researchers to make cloned embryos to get their stem cells, the versatile cells at the centre of a controversy over federal funding.
Scientists believe embryonic stem cells, early master cells that can be coaxed into becoming almost any other cell type, can potentially be used to treat serious diseases.
- REUTERS
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
Wednesday August 1, 2001
Archerd: Hollywood campaigns for stem cell funding
By Army Archerd, Daily Variety Senior Columnist
HOLLYWOOD (Variety) - Not since the Clinton-Gore campaign days has Hollywood been galvanized to support -- not a candidate -- but an issue. The issue: to get President George W. to authorize funds for embryonic stem cell research.
As already noted, congressional support urging government funding has crossed party lines. One of those, a right-to-lifer, who favors the embryo stem cell research funding is Sen. Orrin Hatch (R-Utah).
The Zuckers and fellow Hollywood supporters of this issue have hired a lobbyist in D.C. to fight the fight. The premiere of ``Rat Race'' Monday in Century City raised $420,000, which goes directly to the Juvenile Diabetes Research Foundation's Stem Cell Advocacy Campaign. Paramount underwrote the premiere and the lavish party -- to the tune of $280,000.
``(Paramount boss) Sherry Lansing is fantastic,'' both the Zuckers and Lucy Fisher enthused to me. Janet Zucker added, ``She is a galvanizing force. She has written to President Bush, she is organizing committees and a 'summit conference' in the industry.''
Further, Wick's father, Charles Wick, former head of the USIA under President Reagan, has written to his friends in D.C., including Tommy Thompson, head of the Human Health Services. And Nancy Reagan is also an advocate of the research, having written to President Bush, hopeful her husband's legacy will leave no further suffering for any one afflicted by Alzheimer's, one of the diseases targeted via the research.
Marvin Davis, whose daughter has diabetes, had a conversation with President Bush about the research funding. The Davises are no strangers to the Bush family -- George pere once worked in the Texas oil fields with/for Davis.
Those attending -- and buying party tables at the Las Vegas-style post-premiere screening -- included ``High Rollers,'' ``Big Cheeses'' and ``Fat Cats'' and crossed all political lines. GOP fundraiser Ann Dunsmore got 'em to support the evening's program.
Reuters/Variety REUTERS
Copyright © 2001 Reuters Limited. All rights reserved.
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
Wednesday August 1, 2001
Israelis Turn Human Stem Cells Into Heart Cells
By Will Dunham
WASHINGTON (Reuters) - Scientists said on Wednesday that they had grown heart cells from human embryonic stem cells, an important step toward harnessing the transformational qualities of these primitive master cells to regenerate tissue damaged by cardiac disease.
Israeli researchers said they had created early-stage human heart cells in petri dishes using stem cells derived from an embryo just days after fertilization. They said they envisioned using these cells, after refining the process, to reverse damage inflicted on cardiac muscle by heart attacks.
``It was shown for the first time definitely that the cells that are being created are cardiac cells that show electrical, biochemical and morphological characteristics of early or young cardiac muscle,'' Dr. Joseph Itskovitz-Eldor of the Faculty of Medicine and Rambam Medical Center in Haifa said in a telephone interview.
The publication of the study in the Journal of Clinical Investigation comes as President Bush (news - web sites) considers whether to permit federal funding of research involving human embryonic stem cells.
BODY'S BUILDING BLOCKS
Embryonic stem cells can be viewed as the body's early building blocks. Their wondrous ability to transform themselves into virtually every cell type enables the embryo to grow from a round ball of a few cells into a fully formed body.
Researchers are hoping to use stem cells to create therapies for a variety of illnesses, including heart disease, one of the leading causes of death worldwide.
``The idea is that if you now have a source for heart cells, in the future you can transplant them into a nonfunctioning area (of the heart) and possibly replace the cells,'' said Dr. Lior Gepstein of the Technion-Israel Institute of Technology Faculty of Medicine, an author of the study, along with colleagues including Itskovitz-Eldor and Dr. Izhak Kehat.
``When we have a heart attack, the area of the heart that doesn't receive blood supply actually dies and is replaced by scar tissue,'' Gepstein added. ``Because the adult heart doesn't have any regeneration capacity, this area in the heart won't contract anymore (to pump blood). So this can lead to deterioration in heart function and eventually to heart failure.''
The goal would be to inject the early-stage human heart cells created in the laboratory using stem cells into the damaged area and create healthy cardiac muscle that restores heart function, the researchers said.
Other researchers recently reported they had used mouse stem cells to create mouse cardiac cells.
STANDARD TECHNIQUE USED
The Israeli researchers used human embryonic stem cells to grow an undifferentiated mass of cells using a standard laboratory technique. They then sought to steer the multiplying cells toward transformation into cardiac cells by optimizing the conditions in the petri dish.
As the cells divided, the cells aggregated into microscopic clumps. In about 10 percent of these, the researchers detected small groups of cells that were contracting spontaneously just like cardiomyocytes -- the cells that develop into heart tissue in an embryo.
The researchers then put these groups of cells through a battery of tests to confirm that they were cardiomyocytes and thus destined to differentiate into mature heart cells.
The researchers said the cells checked out in every way, including the genes they activated, the proteins they possessed, their electrical activity as they regularly contracted like a beating heart, their use of calcium and their response to hormones such as adrenaline.
``These are heart cells,'' Gepstein said.
He said the researchers still needed to devise a way to increase the number of these cells produced in the laboratory. Noting that 90 percent of the cells created in the experiment were not heart cells, he said the researchers were exploring different combinations of chemicals to induce the stem cells to produce pure cultures of cardiomyocytes.
The researchers said several million of these cells would be needed for a theoretical transplantation.
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
OTHER ARTICLES ON CLONING AND STEM CELL RESEARCH
Tuesday July 31, 2001
Research Points to Stem Cell Therapy for Diabetes
By Will Dunham
WASHINGTON (Reuters) - Israeli researchers said on Tuesday they had succeeded in coaxing human embryonic stem cells into producing the hormone insulin, in a key step toward creating a revolutionary treatment for type 1 (juvenile) diabetes.
Stem cells that were derived from a human embryo days after fertilization were transformed with chemical prodding into an abundant mass of cells possessing important qualities of the cells of the pancreas that secrete insulin, the researchers said. Those cells are called islet cells, or beta cells.
The findings represent a major stride toward using embryonic stem cells to treat type 1 diabetes. The appearance of the study in the journal Diabetes, published by the American Diabetes Association, comes as President George W. Bush considers whether to allow federal funding for research involving human embryonic stem cells.
Embryonic stem cells are known for their ability to transform into virtually every cell type. Some scientists hope to harness this quality to treat type 1 diabetes by transplanting these cells into the bodies of patients in order to create healthy islet cells to secrete and regulate insulin.
The findings were ``a necessary prerequisite for therapeutic strategies'' for type 1 diabetes using stem cells, the researchers write. The investigators are with the Bruce Rappaport Faculty of Medicine, Technion, Israel Institute of Technology and the Rambam Medical Center in Haifa, and were led by Suheir Assady.
Dr. Christopher Saudek, president of the American Diabetes Association and a professor of medicine at Johns Hopkins University in Baltimore, Maryland, called the findings ''exciting.''
``Up until this point, people have talked about the possibility that human stem cells could be made to produce insulin. But here it is being demonstrated,'' Saudek said in a telephone interview.
In type 1 diabetes, the pancreas fails to pump out insulin due to an immune system attack on its insulin-producing cells. Insulin is a hormone necessary for cells to be able to use blood sugar (glucose), the basic fuel for body cells.
More than 1 million Americans have type 1 diabetes, which strikes children and some adults suddenly, making them dependent on daily insulin injections to stay alive. People with the disease face complications such as heart disease, stroke, amputation, blindness and kidney failure.
Pancreas transplantation is one strategy for combating the disease, but there is an insufficient supply of organs. Investigators are exploring alternative sources of the insulin-producing islet cells.
The Israeli research team said the cells they created in the laboratory possessed many characteristics of islet cells, including insulin production and release. But they acknowledged they had not shown that the cells could regulate insulin secretion based upon the body's glucose levels.
``Although we have not demonstrated glucose responsiveness, we cannot conclude that the cells are glucose unresponsive,'' the researchers write.
``You can say they have demonstrated that you can turn on the gas. What they haven't demonstrated is that you have brakes and accelerators to control it. And that's what you would need in a final use,'' Saudek said.
Because the federal government has never funded research involving human embryonic stem cells, the study could not have been conducted in the United States with grant money from the National Institutes of Health (news - web sites), the major supporter of medical research.
``This is definitely the kind of research that would be accelerated enormously if federal funds could be made available for it,'' Saudek said.
SOURCE: Diabetes 2001;50:1691-1697.
Copyright © 2001 Yahoo! Inc. All rights reserved.
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
Thursday July 26, 2001
Press Release
SOURCE: Union of Orthodox Jewish Congregations of America
Leading Orthodox Jewish Groups Write Bush Encouraging Support For Stem Cell Research With Guidelines
NEW YORK, July 26 /PRNewswire/ -- Today, the Union of Orthodox Jewish Congregations of America and the Rabbinical Council of America wrote to President George W. Bush to express the support of these leading Orthodox Jewish organizations for federal funding for stem cell research so long as such research is conducted under careful guidelines.
The position expressed by the statement was developed in close consultation with leading rabbinic authorities and Orthodox Jewish scientists. The UOJCA represents nearly 1,000 Orthodox Jewish synagogues and the RCA has a membership of over 1,000 Orthodox rabbis.
A complete copy of the letter can be viewed on the Union's website -- http:// www.ou.org, portions are excerpted here:
``[T]he decision you face is one with complex moral dimensions. On the one hand scientific research indicates that there is great life-saving potential in embryonic stem cell research, potential that warrants federal support. On the other hand, we must be vigilant against any erosion of the value that American society affords to human life, including potential human life ... Our Torah tradition places great value upon human life; we are taught in the opening chapters of Genesis that each human was created in G-d's very image. The potential to save and heal human lives is an integral part of valuing human life from the traditional Jewish perspective ... Thus, if embryonic stem cell research can help us preserve and heal humans with greater success, and does not require or encourage the destruction of life in the process, it ought to be pursued.''
``Nevertheless, we must emphasize, that research on embryonic stem cells must be conducted under careful guidelines. Critical elements of these guidelines, from our perspective, relate to where the embryonic stem cells to be researched upon are taken from. We believe it is entirely appropriate to utilize for this research existing embryos, such as those created for IVF purposes that would otherwise be discarded but for this research. We think it another matter to create embryos ab initio for the sole purpose of conducting this form of research ... Other elements of an ethically sensitive oversight regime would include a rigorous informed consent process from future IVF procedure participants, a fully funded and empowered oversight body comprised of scientists and bio-ethicists, and periodic reviews by relevant Executive branch agencies and congressional committees.''
SOURCE: Union of Orthodox Jewish Congregations of America
Copyright © 2001 Yahoo! Inc. All rights reserved.
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
WASHINGTON, July 26, 2001
/U.S. Newswire/ -- In light of the Vatican's statement July 25 that embryonic stem cell research, "even...to help others," is "absolutely unacceptable," a well-known Catholic theologian and an Episcopal priest, both affiliated with the Religious Coalition for Reproductive Choice have expressed other views on the moral issues involved.
According to theologian Mary E. Hunt, Ph.D., and Episcopal priest The Reverend Dr. Katherine Hancock Ragsdale, prohibiting such research -- or federal funding for research -- elevates respect for embryos slated for destruction above respect for those who could benefit from research findings.
"As people of faith we are called to be partners with God in healing and in the alleviation of human pain and suffering," they said. "Our respect for life includes respect for the embryo and fetus. Therefore, decisions about research should be made responsibly, under existing federal guidelines."
Pope John Paul II's condemnation of using embryonic stem cells for research represents the view of those who see fetal and embryonic life as morally equivalent to human persons. Most Americans do not. The question before President Bush is research on excess embryos created for in vitro fertilization and slated for destruction.
The enormous life-saving potential of embryonic stem cells cannot currently be matched by adult stem cells, according to researchers. Narrow theological arguments disregard a basic religious principle, that of the common good of all humanity. "The Pope is only one religious voice in the debate," they said. "Many other faiths and individuals have strongly held beliefs in support of this research. We hope President Bush will listen to all sides of the issue."
The Religious Coalition for Reproductive Choice, founded in 1973, is the national alliance of pro-choice religious organizations.
Copyright © 2001 Yahoo! Inc. All Rights Reserved.
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
Wednesday July 25, 2001
Bone Marrow Cells Can Develop Into Kidney Tissue
By Richard Woodman
LONDON (Reuters Health) - Scientists said on Wednesday they had discovered that adult stem cells found in the bone marrow are capable of turning into kidney cells after a bone marrow transplant. The discovery, made in mice and human transplant patients, suggests that bone marrow-derived cells could be used to treat kidney failure, although more research is needed to determine if this is true.
The stem cells found in bone marrow are immature cells that can give rise to all cells of the blood and immune system. Past studies have shown they also have the potential to transform into liver cells and researchers in London said their work proves for the first time that these remarkably plastic cells can also transform themselves into kidney cells.
They hope that their findings, published in the online version of the Journal of Pathology, could one day enable doctors to restore kidney function to patients suffering from kidney failure or perform gene therapy for kidney diseases.
The team, from the Imperial Cancer Research Fund and Imperial College School of Medicine in London, examined kidneys of female mice that had received a male bone marrow transplant and kidney biopsies from eight male patients who had received kidney transplants from female donors.
By using a special DNA probe to detect the sex chromosomes, the researchers showed that circulating stem cells frequently engraft into the kidney and differentiate into kidney tissue cells.
They said bone marrow-derived cells were found in both normal mouse kidneys and in the human transplanted kidneys.
``These data indicate that bone marrow cells contribute to both normal turnover or renal epithelia and regeneration after damage, and it is suggested that this could be exploited therapeutically,'' they state in the journal.
Professor Malcolm Alison, a research pathologist working at both Imperial Cancer and Imperial College School of Medicine, said in a news release, ``Our laboratory experiments showed the presence of cells in the kidney that came from bone marrow stem cells, but we then went a step further and proved it can actually happen in the human body.''
According to Dr. Richard Poulsom, lead author and research pathologist at Imperial Cancer Research Fund, ``The potential for advances in medicine from using adult stem cells is enormous. They can give rise to many different types of cells so any organ may one day be repaired. Using adult stem cells also avoids the ethical dilemmas associated with embryonic stem cell work.''
Stem cells can also be collected from embryos and are thought to have great potential in treating adult illnesses, but are currently at the center of a raging debate over the ethics of using such tissue. While adult stem cells are promising as well, many researchers believe that embryonic cells can form an even greater variety of adult body tissues.
Sir Paul Nurse, Director General of Imperial Cancer Research Fund, said, ``This is a great achievement and shows real progress by the team which, almost exactly a year ago, discovered that bone marrow stem cells are capable of turning into liver cells and repopulating damaged liver.''
SOURCE: Journal of Pathology 2001;193:1-7.
Copyright © 2001 Reuters Limited. All rights reserved.
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
Wednesday July 25, 2991
Vatican Says No Exceptions to Pope's Stem-Cell Stand
Bush Returns to White House - (WKMG, Orlando)
By Philip Pullella
VATICAN CITY (Reuters) - The Vatican (news - web sites), rejecting suggestions that Pope John Paul (news - web sites)'s stem-cell warning to George W. Bush left the president room to maneuver, said on Wednesday he condemned all research using embryos without exception.
In a public address to Bush at their meeting on Monday, the Pope referred to stem-cell research, a topic at the forefront of the conservative president's mind as he deliberates whether to permit federal funding for such work.
Stem cells can be taken from embryos and are capable of developing into cell types that make up the body. Scientists believe they can lead to cures for diseases such as Alzheimer's, Parkinson's, diabetes, spinal cord injuries and cancer.
The Pope defined as evil ``proposals for the creation for research purposes of human embryos, destined to destruction in the process.''
While the Pope clearly referred to embryos created to be destroyed by research he did not specifically refer to those created for use in in-vitro-fertilization (IVF) and which are left over after a woman becomes pregnant.
The Washington Post on Tuesday cited U.S. administration officials as saying the Pope's words were ``sufficiently ambiguous'' to provide comfort for Bush if he approves funding for stem-cell research with extra embryos in fertility clinics.
The paper said this was a compromise the conservative president was considering, adding that the U.S. administration believed the Pope's words could have been much broader if he wanted to include IVF embryos.
The Vatican, anxious to cut short any suggestion of different rights for different embryos, weighed in firmly with an official statement by its chief spokesman.
Joaquin Navarro-Valls quoted from the Pope's 1995 encyclical Evangelium Vitae (The Gospel of Life), which specifically condemned the destruction of embryos produced for IVF.
``This moral condemnation also regards procedures that exploit living human embryos and fetuses -- sometimes specifically 'produced' for this purpose by in-vitro fertilization -- either to be used as 'biological material' or as providers of organs or tissue for transplants in the treatment of certain diseases,'' the statement said.
``The killing of innocent human creatures, even if carried out to help others, constitutes an absolutely unacceptable act,'' it said.
STEM CELLS ARE HOPE FOR FUTURE
Stem cells can give sick people new cells that have been instructed to act as replacements for damaged tissue.
They can also be taken from organs and tissues in a body -- these are known as adult stem cells -- but scientists say that research on embryonic cells is more promising.
The Church, which teaches that life begins at the moment of conception, does not oppose adult stem-cell research.
After his meeting with the Pope, Bush said he would keep the Pontiff's stand in mind when making his decision on whether federal money should be used for embryonic stem-cell research.
Bush, who is opposed to abortion except in cases of rape and incest, is said to be agonizing over the stem-cell decision, one of the most important of his presidency.
``I do care about the opinion of people, particularly someone as profound as the Holy Father,'' he said on Monday.
``I'll take that point of view into consideration as I make up my mind on a very difficult issue confronting the United States of America. It's the need to balance value and respect for life with the promise of science and the hope of saving life,'' he said.
Copyright © 2001 Reuters Limited. All rights reserved.
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
Monday July 23, 2001
Pope Warns Bush of 'Evil' of Embryo Cell Research
President Bush to Meet Pope for First Time - (WKMG, Orlando)
By Philip Pullella
CASTELGANDOLFO, Italy (Reuters) - Pope John Paul (news - web sites) warned President Bush (news - web sites) on Monday of the ``evils'' of stem-cell research using embryos and spoke of the right to life and dangers of globalization.
Bush, 55, dressed in a dark suit, appeared slightly nervous as he was introduced to the 81-year-old Pope, who greeted him while leaning on a cane.
Bush, asked later about his feelings during the meeting, said: ``It's hard to describe. I'm not poetic enough to describe what it's like to be in his presence.''
Meeting the Pontiff for the first time as president, Bush took the ailing Pope's left hand and helped him walk to chairs where they held 35 minutes of private talks in a room overlooking a lake at the papal summer residence south of Rome.
``This is where the Popes spend their summers,'' the Pope said while photographers clicked away.
``Yes Sir. And I can understand why. It's so beautiful here,'' Bush said.
During their private talks, which Bush later described as a ''good discussion,'' the two men spoke of the recent violence-marred Group of Eight summit in Genoa, the Middle East, and ways of helping the developing world, a Vatican (news - web sites) source said.
PUBLIC SPEECH
The Pope saved his thunder for his public speech.
In a talk woven around the theme of respect for life, the Pope made a clear reference to stem-cell research, a topic in the forefront of the conservative president's mind as he deliberates whether to permit federal funding for such work.
He spoke of ``evils such as euthanasia, infanticide and, most recently, proposals for the creation for research purposes of human embryos, destined to destruction in the process.''
The Catholic Church condemns stem-cell research using embryos because they are destroyed in the process but does not oppose other forms of stem-cell research where cells are taken from body tissues and life is not threatened.
The Pope's specific words on stem-cell research concerning embryos appear to leave maneuvering room for Bush as he ponders what kind of stem-cell research to approve for federal funding.
Bush later told a news conference he would keep the Pope's words in mind when thinking about his decision.
``I do care about the opinion of people, particularly someone as profound as the Holy Father,'' he said.
``I'll take that point of view into consideration as I make up my mind on a very difficult issue confronting the United States of America. It's the need to balance value and respect for life with the promise of science and the hope of saving life,'' he said.
Advocates believe research with embryonic stem cells, the early master cells formed soon after a human egg is fertilized, could lead to medical advances.
The aging Pontiff also spoke out, albeit indirectly, against the death penalty, which Bush supports.
``A free and virtuous society, which America aspires to be, must reject practices that devalue and violate human life at any stage from conception until natural death,'' he said.
During Bush's six years as Texas governor, the state carried out 152 executions, the highest rate in the United States. Under his presidency, U.S. federal authorities resumed executions after 38-years. Two men have been put to death.
POPE WARNS OF DOWNSIDE OF GLOBALISATION
Saying ``a global world is essentially a world of solidarity,'' the Pope decried that many do not reap globalization's benefits.
``The Church cannot but express profound concern that our world continues to be divided, no longer by the former political and military blocks, but by a tragic fault line between those who can benefit from these opportunities and those who seem cut off from them,'' he said.
He also repeated calls for industrialized countries to share technology with poorer countries, to respect the environment, to cancel or significantly reduce developing country debt and to be open to immigrants.
In his speech, Bush avoided controversy, praising the Polish Pope for his role in defense of human rights and referring to his role in the fall of communism in Eastern Europe.
``You have urged men and women of goodwill to take to their knees before God and to stand, unafraid, before tyrants...this has added greatly to the momentum of freedom in our time,'' Bush said
After the private talks, Bush's wife Laura and daughter Barbara, both clad in black and wearing mantillas for the occasion, were called in and shown the view from the papal balcony, overlooking Lake Albano, an extinct volcano.
Copyright © 2001 Reuters Limited. All rights reserved.
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
WHERE SCIENCE IS TAKING US
Report: Test-Tube Baby Born to Save Ailing Sister
Reuters
WASHINGTON (Oct. 3, 2000) - A Colorado couple used genetic tests to create a test-tube baby that would have the exact type of cells desperately needed to save their 6-year-old daughter, the Washington Post reported on Tuesday.
The newspaper said it was the first time a couple was known to have screened their embryos before implanting one in the mother's womb for the purpose of saving the life of a sibling.
The baby, named Adam, was born in Denver on Aug. 29. Doctors collected cells from his umbilical cord, a painless procedure, and on Sept. 26 infused them into his sister Molly's circulatory system, according to the Post report.
Molly suffered from an inherited bone marrow deficiency that is universally fatal without a transplant like the one she received from her newborn brother. She is recuperating in a Minneapolis hospital, and within about a week doctors should know whether the procedure was successful, the Post said.
The procedure is both a promising and worrisome harbinger of where scientific advances are taking human reproduction in the near future -- at least for those who can afford to take that path, the Post said, quoting doctors and ethicists.
''We knew we were running out of time,'' Charles Strom, director of medical genetics at Illinois Masonic Medical Center, where the genetic testing was done, told the paper.
Now, he said, the girl has an 85 percent to 90 percent chance of being largely free of the marrow disease.
But the case also raises questions about parents' ability to choose the traits of their children, for whatever practical -- or capricious -- reason they may have.
GIRL FACED RARE, DEADLY DISEASE
The girl who received the cell transplant, Molly Nash of Englewood, Colorado, was born with Fanconi anemia, an inherited disorder that causes a massive failure of bone marrow cell production.
Children with the disease suffer from anemia, bleeding disorders and severe immune system problems and generally die from leukemia or other complications by the time they are 7.
The only effective treatment is to get a batch of healthy cells from a perfectly matched sibling to replace the ailing child's faulty bone marrow cells.
The Post said Molly's parents, Lisa and Jack Nash, had long wanted more children, but were afraid to conceive another for fear that any new baby would be affected by the disease. Each parent carries both a normal and a faulty version of the Fanconi gene, which meant they had a 25 percent chance of having an affected child with each pregnancy.
But a few years ago they learned of a new technique under development called pre-implantation genetic diagnosis.
Researchers harmlessly pluck a single cell from embryos that have been created by standard in vitro fertilization and are developing in a laboratory dish. They test each of those cells for the presence of a disease gene, and then transfer to a woman's uterus only those embryos whose cells test normal.
Lisa Nash told the Post that when she tried in vitro fertilization for the first time a few years ago she turned to pre-implantation genetic diagnosis primarily to select an embryo unaffected by Fanconi. She allowed researchers to test the embryos for compatibility with Molly's cells, but the tests were not very good then, she said.
The couple went through this four times, and each time benefited from increasingly sophisticated cell typing tests. But none of the attempts resulted in a pregnancy.
Finally, in the fertility cycle initiated late last year, two of the couple's 15 embryos tested as both free of the disease and having a perfect match for Molly, Strom said.
Only one of those two embryos was healthy enough to transfer to Lisa Nash's womb. On Christmas Eve the
couple learned that the embryo had implanted properly and that Lisa Nash was pregnant, according to the Post report.
After Adam was born, doctors saved blood cells from his umbilical cord. Research has shown umbilical cord cells, like transplanted bone marrow cells, can go to a recipient's bone marrow and repopulate the marrow space with healthy cells.
The transplant was performed last week at Fairview-University Hospital in Minneapolis, which specializes in bone marrow replacements for children with Fanconi anemia.
''Molly was holding Adam in her lap'' while the cells dripped through a plastic tube into the girl's chest, Nash told the Post. ''It was the most awesome, monumental experience of our life, yet it was so simple. You'd think there'd be thunderbolts and lightning, but it was calm.''
So far, doctors said, Molly is doing well.
University of Minnesota cord blood specialist John Wagner, who oversaw the transplant, told the Post there had been no complications, and Molly would soon be out of the high-risk period that follows such procedures. Doctors suppressed her immune system with the radiation and chemotherapy to increase the chances the cells would not be rejected.
Copyright 2000 Reuters Limited. All rights reserved.
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
Opposition to human cloning will 'blow over,' scientist says
January 7, 1998
Same technique used in sheep cloning
Ethical, legal dilemma
CHICAGO (CNN) -- A scientist who plans to clone babies for infertile couples believes any opposition to his work will be short-lived. "I think it will blow over," Richard G. Seed told CNN on Wednesday.
"There were an awful lot of people against the automobile, too," he said in a live interview by telephone from Chicago. "Any new technology ... creates fear and horror." But as time passes, human cloning will receive "enthusiastic endorsement," he said.
CNN's Dr. Steve Salvatore explains the cloning dilemma
Seed, who has a Harvard doctorate in physics and has done fertility research in the past, plans to begin his work on a human clone within three months . "My target is to produce a two-month pregnant female (within the next 18 months)," he said.
If he is barred from pursuing his work in the United States, Seed said he plans to go to another country. He said he has talked with officials in Tijuana, Mexico, and also was considering the Cayman Islands and the Bahamas.
In a separate interview with National Public Radio (NPR) correspondent Joe Palca, , Seed said it was his objective to set up profitable human clone clinics, first in the Chicago area and then at "10 or 20 other locations" in the U.S. and "maybe five or six" internationally.
~~~~~~~~~~~~
'Cloning and the reprogramming of DNA is the first serious step in becoming one with God.'
— Scientist Richard G. Seed
Seed, who is not a medical doctor, says he has already assembled a group of doctors willing to work with him and has four couples who have volunteered to be cloned.
The goal for each of them is to achieve a pregnancy within a year and a half.
The researchers will use a donor cell from either the mother or father and test it for genetic abberations, he said.
He declined to name any of the couples and it was not immediately clear where in the Chicago area Seed planned to open his proposed clinic. He told USA Today he needs $2 million to begin his privately funded project but has only raised "a few hundred thousand" dollars.
Same technique used in sheep cloning
Human cloning goes under the microscope
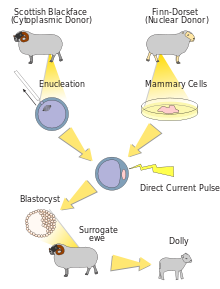
Seed plans to use the same technique utilized by Scottish scientists in 1996 to clone the adult sheep Dolly, the first mammal cloned from adult tissue.
The human cloning procedure involves taking an unfertilized egg from a female, removing the nucleus, which contains most of the genetic information, and replacing it with the nucleus of a cell from the person to be cloned.
The hard part is tricking this egg into acting as if it has been fertilized by a sperm, thus starting it dividing as if it were a new baby, instead of just creating more skin cells or liver cells or cells of whatever organ the nucleus was taken from.
If the technique is successful, the fertilized egg would grow to 50 to 100 cells and the embryo would then be transferred to a woman. A baby clone would be born nine months later.
Seed says the cloned babies he and his colleagues would create would have no chromosomal damage and a normal life span.
He first talked about his plans December 5 at a little-noticed Chicago symposium on reproductive technologies sponsored by the Illinois Institute of Technology, Palca told CNN.
Ethical, legal dilemma
Seed will attempt to use the same techniques used to clone Dolly
After Dolly was cloned, President Clinton set up an advisory group which recommended last year that Congress pass a law making human cloning illegal.
Harold Shapiro, who headed the panel -- the National Bioethics Advisory Committee -- believes Seed's project is "scientifically and clinically premature," with many legal and ethical issues yet to be resolved.
In the future, however, Shapiro acknowledged there might be cases where human cloning could benefit infertile couples.
Clinton issued an executive order blocking the use of federal funds on human cloning research and proposed a law banning such research for five years, actions Seed disagrees with. "I am an independent thinker," he told NPR.
Several measures to ban cloning are awaiting action in Congress. In the meantime, the Food and Drug Administration says it has the authority to regulate human cloning research.
The American Medical Association said Wednesday it does not have an official policy on the subject but expects members to discuss the issue at a meeting in June.
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
Human Cloning Plans
January 6, 1998 -- American scientist Richard Seed tells NPR’s All Things Considered that he and his colleagues are set to begin work on human cloning within the next 90 days. In an interview, NPR’s Science Correspondent Joe Palca reports that Seed and some Chicago-area doctors have the skills and equipment to make an attempt at the procedure, which they plan to offer to infertile couples wishing to have children.
The controversial move runs counter to President Clinton's directive that the public and private sectors refrain from human cloning research. The president made that request last year in the wake of news that Scottish scientists successfully cloned the adult sheep Dolly. The prospect of human cloning has been widely denounced by ethicists, scientists, and public officials.
But, as Palca reports, Seed sees human cloning as a moral imperative -- something that will bring humankind closer to God. Hear Richard Seed explain his rationale.
Ethicist and lawyer John Robertson disagrees with Seed’s rationale but says some people may find legitimate uses for human cloning in the future. Listen to Robertson's arguments.
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
The Lancet, Jan 2, 1999
Human cloning trial met with outrage and scepticism. Kelly Morris; Jonathan Watts.
The announcement last month by South Korean scientists that they had made the world's first human clone sparked public outrage and scepticism among scientists.
Researchers at Seoul's Kyunghee University Hospital infertility clinic reported on Dec 16 that they had fused an adult human nucleus with an enucleated egg to create the embryo, which divided twice to reach the four-cell stage. The experiment was stopped on Dec 11 to avoid contravening ethical guidelines, but team leader Lee Bo-Yeon said the next stage would have been to transfer the embryo to a womb. "If implanted into the uterine wall of the carrier, we can assume that a human child would be formed and that it would have the same gene characteristics as the donor", he told reporters.
Lee explained that the team had used the so-called Honolulu technique. This method, which was used to clone mice in Hawaii earlier last year, involved insertion of a somatic-cell nucleus into an enucleated egg followed by activation of the egg. This was a modification of the technique used at the Roslin Institute (Roslin, UK) to create Dolly, the first vertebrate cloned from an adult cell. In Dolly's case, a whole somatic cell was inserted into an enucleated egg, and the two cells were simultaneously fused and activated by passage of a small electric current.
Lee's claims were dismissed as unsound by scientists around the world, many of whom noted that Lee's unpublished work is not part of a major cloning research programme. Yukio Tsunoda (Kinki University, Nara, Japan), the first scientist to clone twin calves, said, "I have never heard of such an experiment taking place and at the moment I don't believe it is true".
Harry Griffin, a scientific director at the Roslin Institute, said that "taking a putative embryo to the four-cell stage doesn't actually tell us anything at all", since a fertilised human egg is "preprogrammed" to divide to at least the eight-cell stage. To produce a live, healthy clone, the 100 000, mainly silent, genes in the somatic-cell nucleus need to be activated rapidly and "perform perfectly for the next 9 months", Griffin explained. The generally low success rate with cloning sheep, calves, and mice suggests that a cloned human embryo would probably be stillborn or die soon after birth. "Nuclear transfer is not in-vitro fertilisation with a twist."
Another risk associated with cloning from somatic cells is the potential for inheritance of somatic-cell mutations from the donor. Nuclear transfer bypasses mechanisms that correct DNA errors in germ cells so cancer incidence may be increased in clones. Scientists are also concerned that the DNA in a cloned animal may behave like that from an animal that has the combined age of the donor and offspring, and that this might shorten the clone's lifespan.
These scientific concerns added to the serious ethical reservations expressed internationally as the world came face-to-face with the possibility of human cloning. Protesters demonstrated outside Kyunghee University Hospital demanding that Lee and his team apologise to the human race for their "inhuman experiment". Local newspapers reacted with a mixture of pride and dismay, though there was widespread agreement that South Korea needs new laws to curb cloning research. Currently, the only restraint is voluntary--a 1993 resolution by the South Korean Medical Association.
Lee said he would continue his studies, but only within the limits of the law. "I understand the concerns of people who are opposed to it."
~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~
The New Republic, March 1, 1999 p20(1)
Will Homo sapiens become obsolete?: MEDICAL EVOLUTION. (stem-cell research raises cloning controversy) Gregg Easterbrook.
Abstract: Two scientists have isolated embryonic stem cells; the cells that are the precursor of all body parts. Also, they have induced them to copy themselves into like cells that can generate any type of human tissue. The research uses tissue from aborted fetuses, and this aspect of it has raised some controversy. Also, some fear this stem-cell research puts biotechnologists only a step away from human cloning. The 2 scientists who pioneered the research are calling on Congress to lay down clear guidelines for the field.
For John Gearhart, a biologist at Johns Hopkins University, professional life had been an exercise in slamming against walls. Gearhart's specialty is Down's syndrome, triggered when one of the infant body's chromosomes copies itself once too often. Gearhart had spent 20 years trying to puzzle out this genetic error. "All our data suggested that Down's was caused by something that happens quite early in embryo genesis," he says--but the only way to find out what happens then would be to conduct experiments on human embryos, a prospect repugnant at best. Trying to think his way out of the problem, Gearhart wondered; What if there was a way to isolate and culture embryonic "stem cells," the precursors of all body parts? If they could be transferred to the laboratory, it might become possible to study the cytology of conception.
Stem cells are the philosopher's stones of biology, magical objects capable of metamorphosing into any component of the body: heart, nerves, blood, bone, muscle. Mammal embryos begin as a clump of stem cells that gradually subdivides into the specific functional parts of the organism. Researchers have long assumed that, because stem cells are genetically programmed to change into other things, it would never be possible to control them, let alone culture them. But Gearhart and another researcher working independently, James Thomson of the University of Wisconsin, found this is not so.
Three months ago, Gearhart and Thomson announced that they had each isolated embryonic stem cells and induced them to begin copying themselves without turning into anything else. In so doing, they apparently discovered a way to make stem cells by the billions, creating a biological feedstock that might, in turn, be employed to produce brand-new, healthy human tissue. That is, they discovered how to fabricate the stuff of which humanity is made.
Researchers had already demonstrated that stem cells might be a medical boon by showing that such tissues extracted from aborted fetuses could reverse symptoms of Parkinson's disease. But so many fetuses were required to treat just one patient that the technique could never be practical, to say nothing of its harrowing character. By contrast, Gearhart and Thomson have found that stem cells can be reproduced roughly in the way that pharmaceutical manufacturers make drugs.
If researchers can convert stem cells into regular cells like blood or heart muscle and then put them back into the body, then physicians might cure Parkinson's, diabetes, leukemia, heart congestion, and many other maladies, replacing failing cells with brand-new tissue. Costly afflictive procedures such as bone-marrow transplants might become easier and cheaper with the arrival of stem-cell-based "universal donor" tissue that does not provoke the immune-rejection response. The need for donor organs for heart or liver transplants might fade, as new body parts are cultured artificially. Ultimately, mastery of the stem cell might lead to practical, affordable ways to eliminate many genetic diseases through DNA engineering, while extending the human life span. Our near descendants might live in a world in which such killers as cystic fibrosis and sickle-cell anemia are one-in-a-million conditions, while additional decades of life are the norm.
Granted, sensational promises made for new medical technologies don't always come to pass, and some researchers are skeptical about whether stem-cell technology will pan out. But Harold Varmus, head of the National Institutes of Health (NIH), recently declared, "This research has the potential to revolutionize the practice of medicine." Notes John Fletcher, a bioethicist at the University of Virginia, "Soon every parent whose child has diabetes or any cell-failure disease is going to be riveted to this research, because it's the answer." Ron McKay, a stem-cell researcher at the National Institute of Neurological Disorders and Stroke, says, "We are now at the center of biology itself." Simply put, the control of human stem cells may open the door to the greatest medical discovery since antibiotics.
But there are disquieting aspects to stem-cell research, too. The first is that, for now, the only way to start the process of controlled stem- cell duplication is to extract samples from early human life. Gearhart used fetuses aborted by Baltimore women; Thomson, embryos no longer wanted by Wisconsin in vitro fertilization (IVF) clinics. Gearhart, Thomson, and other stem-cell researchers propose to continue drawing on such "resources," as some bloodless medical documents refer to the fetus and the embryo. This is possible because, even though Congress has placed a moratorium on federal funding for experimentation on most IVF embryos and most kinds of fetal tissue, no law governs what scientists can do to incipient life using private funding, either in research settings or within the burgeoning IVF industry.
Because the rules have banned embryo research by federally funded biologists, but not comparable private science, Congress has created the preposterous situation in which most stem-cell research is not being done by publicly funded scientists who must pass multiple levels of peer review and disclose practically everything about their work. Instead, most stem-cell science is in the hands of corporate-backed researchers. Gearhart's and Thomson's projects, for example, are being underwritten by Geron, a company whose name derives from "gerontology," and which anticipates a licensing El Dorado if stem-cell-based good health can be patented and sold to the seniors' market. "That a sensitive category of research is legal for people who are not publicly accountable, but illegal for those who are accountable, is just very strange," says Thomson.
But the greatest anxiety about stem-cell research is that it will make human cloning respectable. Many of the techniques being perfected for the medical application of stem cells are just a hop, skip, and a jump away from those that could apply to reproductive cloning. Society isn't even close to thinking through the legal, ethical, regulatory, and religious implications, but, thanks to stem-cell research, cloning may arrive much, much sooner than anyone expects.
Stem cells stand in the vanguard of human life. When a sperm penetrates an egg, it triggers a majestic sequence whose first step is to create a new structure that is composed mainly of stem cells. Biologists call such cells "undifferentiated," meaning they have not yet decided what they will be. Once the fertilized ovum implants in the uterus, differentiation starts. Some stem cells become placenta; others begin differentiating into the baby's organs, tissue, or blood. A stem cell might divide into any of the many components of the body, but, once it does, it can only continue growing as that part.
Because once a stem cell begins to differentiate it cannot turn back, biologists assumed that all stem cells could never turn back. But, in 1981, experimenters succeeded in extracting stem cells from the embryos of mice. By the mid-'90s, researchers had learned which chemicals instruct mouse stem cells to become particular tissue types and how to insert the new tissues back into mice. Loren Field of Indiana University became so adept at signaling mouse stem cells to become mouse heart cells that "his lab is almost pulsating with heart cells beating in dishes," Gearhart says.
But, though the stem cell clearly dominates human germination, mysteries abound. The overriding enigma is why stem cells work so phenomenally well in the womb but then stop working when the body matures. After all, every cell in the body contains a complete DNA blueprint for a person's being. In theory, if your coronary arteries became clogged, your DNA could direct the creation of stem cells that would subdivide into new arteries to replace the failing tissue. In some lower animals that regrow limbs, this is roughly what happens. But, after reaching maturity, the human body never again draws on its DNA blueprints to replace tissue more complex than skin. Thomas Okarma, vice president for research for Geron, asks, "Who is really more highly evolved, the lizard that can grow new body parts, or us?" Using stem cells to make fresh new tissue or organs, as if the body was still nascent, is the revolutionary therapy Geron hopes to market.
Another mystery concerns the regulator that shuts stem-cell division off, called the telomere. Any human cell is capable of dividing roughly 50 times; after that, growth stops. Cells are born with a length of telomere, which gets slightly shorter each time the cell divides. When the last snippet is gone, after about 50 divisions, the cell can no longer duplicate. Running out of telomeres is one reason we age.
Why God or natural selection put telomeres into the cell is hotly debated among theorists. But, along with their other dizzying advances, stem-cell researchers have learned to rejuvenate the telomere, at least in the lab. Using an enzyme called telomerase, Geron scientists have already guided batches of stem cells through 200 divisions, with the descendant tissue being stable and nonmalignant. Researchers joke that cells that endlessly divide are "immortal," but this has always been a sardonic adjective, since the only immortal cells observed in natural biology are cancerous. Now Geron is making hale, normal cells that don't grow old, and wondering if this effect can be transferred to people. Therapeutic use of "immortal" cells would not confer unending life (even people who don't age could die in accidents, by violence and so on) but might dramatically extend the life span. Stem-cell research is spinning out so many breakthroughs so fast that its pursuit of the ageless cell, which in other contexts might be the scientific story of the century, here seems like just an asterisk.
The hope among stem-cell researchers is that practical treatments will be available in ten years or less. Biologists have been encouraged by the reasonable success of stem cells collected from the umbilical cords of newborns and used to treat conditions such as bone-marrow disease. (The sudden awareness that stem cells can be manipulated is causing crash programs to collect umbilical cords, which are rich in these enchanted cells.) But many things could go wrong between here and a new form of medicine. Loren Field's experiments with mouse hearts, for example, have shown that 97 percent of artificially cultured, stem- cell-based heart tissue works properly when transplanted into mice. But for reasons unknown, the other three percent becomes malignant. Obviously that problem has to be surmounted before human stem-cell trials can be contemplated.
More pressing is resolving the basic ethics of stem-cell research. When Gearhart first proposed to use aborted fetuses as the basis of his stem-cell research, his sponsoring university was not thrilled. Although research on aborted fetuses is legal, it's a gray area, and restrictions are many. Occasionally, a maverick researcher protests this situation by asking the discomfiting post-Roe question: If it's okay to terminate a fetus, why isn't it okay to experiment on the remains? Few object to research on the cadavers of adults. Do we ban research on the aborted out of a sense of guilt that we should not add mutilation to the wrongs suffered by a life denied?
Gearhart's stem-cell extractions employ "fetal tissue," which researchers may view as differing from fetuses, though the reasoning is obscure. Johns Hopkins put his proposal through eleven levels of legal and ethical review, and also review by the university's security office, which worried that the degenerate fringe of the pro-life movement would target the project. Gearhart chose to focus on a type of stem cell called a "primordial germ cell," which he extracts from a region on the aborted fetus called the "urogenital ridge." Gearhart will work with fetuses no more than nine weeks old. Around nine weeks is when the fetus begins to appear recognizably human, though it is believed there is no consciousness until weeks later. He says, and not without defensiveness, "Remember, it's not criminal at this point." Technically, Gearhart's project might have qualified for federal funding, but he never asked, knowing there would be an outcry.
For his part, Thomson isolates stem cells from the "inner mass" of roughly week-old embryos that were conceived in IVF clinics but which couples elected not to use. Thomson thus extracts tissue from the early, pre-uterus stage in which it is impossible to tell which stem cells will become the placenta and which the person. Strictly speaking, then, Thomson has no way of knowing whether the cells he plucks could have grown or would have been lost anyway. Though Thomson's extractions are timed to occur at what seemed to him the least ethically perilous moment of incipient life, in the cockeyed world of bioethics regulation, his recent experiments clearly could not have qualified for federal funding, since he was employing embryos.
Both Gearhart and Thomson call on Congress to enact clear legal guidelines for their field. Thomson says, "The human embryo is the most special cell in biology, and there are just some things you shouldn't do to embryos"--mainly clone them. The primary point stem-cell researchers make in their own favor is that the cells they experiment upon, once brought into the lab, might be made into muscle or blood, but can no longer become a human being. This assertion seems true, though slightly cute, since the reason the cells cease being capable of personhood is that they've been artificially snatched from it. But then no one plans to conceive the IVF embryos that Thomson gets, and the fetuses Gearhart receives have already had their lives terminated. Neither biologist can change these things, though both might change others' lives for the better.
Reflecting the delicacy of the situation, stem-cell researchers are
beginning to wrestle over the terms "totipotent" and "pluripotent." A totipotent cell is what exists at the earliest germination stage, when each stem cell is capable of becoming a whole person. A pluripotent tissue is an isolated stem cell, capable of transforming into any desired cell type, but not of becoming a whole person. Not, at least, with current technology.
In January, government lawyers sided with the pluripotent versus totipotent distinction, ruling that the NIH can begin funding stem-cell research on the grounds that the cells being worked with cannot become persons and thus are not embryos. This ruling hasn't yet taken effect; assuming it does, there will be beneficial results. Publicly funded scientists from research-center universities will jump into stem-cell investigations: research-center scientists are generally the country's best, and always the most accountable. Equally important, federal funding will move stem-cell findings into the public domain rather than allowing them to become proprietary. Geron shares samples of its stem cells with academics but asks the recipients to sign a statement that Geron owns the knowledge embodied in the cell line. Once public funding flows, proprietary claims will diminish.
Bringing public funding to stem-cell research will force a public debate on this new biology. There has been little so far. In Congress, a few members, such as Representative Jay Dickey of Arkansas, have declared themselves opposed, for pro-life reasons, to any research on embryonic cells. A few members, such as Senator Tom Harkin of Iowa, have openly endorsed stem-cell studies. Senator Arlen Specter of Pennsylvania was expected this year to introduce legislation making human stem-cell research explicitly legal, as it is in the United Kingdom. But Specter now says he will postpone action, feeling the time isn't right.
Many researchers were pleased by this decision, fearing that drawing public attention to today's stem-cell research might only provoke a backlash, whereas, once treatments derived from stem cells are available, lobbying impetus for the technology will be unstoppable. But, while the science world may be trusted to police its own for professionalism and high standards, it can't be trusted with the ethical end of this debate.
Science types tend to be unreflective boosters of funding, funding, and more funding. A 1994 NIH commission on whether to allow embryonic research endorsed it without meaningful objection, tossing off slighting remarks--like a statement that the human embryo is "significantly smaller than the period at the end of this sentence," as if that had anything to do with anything. Stem-cell research came over the horizon so fast that the 1994 panel's work quickly became obsolete, and now a National Bioethics Advisory Commission has been convened to study the latest developments. But it, too, seems mainly concerned with rationalizing the status quo. In January, I watched as the bioethics commissioners had a chance to grill Gearhart and Thomson. The commissioners were awestruck and deferential, asking what they could do to help win stem-cell funding--not much more aggressively than Dolly the cloned sheep herself might have. Toward the end, one commissioner, Rhetaugh Dumas of the University of Michigan, asked the sole unfriendly question: Does stem-cell research have any downsides? When neither of the scientists spoke, Dumas looked around at her fellow commissioners and asked skittishly, "Was that an inappropriate question?" It wasn't a reassuring moment.
One reason the interplay between science and religion has become a topic again is that thoughtful researchers know biotechnology is raising questions science can't answer. For one, whether stem-cell research is ethically good, bad, or indifferent depends largely on when life begins, and on this subject there is neither definitive science nor ethics. Many Catholic, evangelical, and Islamic theorists say that life begins at the moment sperm meets egg; therefore, a single cell can have sacred rights. But this seems not quite right, because DNA sets from egg and sperm do not immediately merge; the ovum divides once before the onset of genetic recombination. Besides, what would happen if cloning rendered every cell in the body a potential person? Varmus, the NIH director, has asked, "If we say any cell has the potential to be a human being, then every time you cut your finger, do you have to wear black?"
Obstetricians sometimes mark the big moment as the time, around two weeks after conception, when the fertilized egg implants in the womb and pregnancy begins. Thomas Aquinas taught that the fetus became "ensouled" after about six weeks. This view, which was Vatican policy until 1869, differs only somewhat from Gearhart's conclusion that fetal tissue extractions should stop after about nine weeks. Roe declared that the rights of life are in view when the fetus is capable of existing outside the mother, a milestone technology keeps pushing back toward the second trimester. Surely once brain activity begins, around the twenty-fifth week, the fetus has become human, leaving the late- term abortion a cause for moral abhorrence, except when the life of the mother is threatened. But, before that point, it's all speculation.
Consider that huge numbers of embryos die during the normal process of conception. As many as a third of pregnancies end in natural miscarriage, even with the best obstetric care. Studies increasingly show that it is surprisingly common, even in the absence of contraceptives, for eggs to be fertilized but never begin to divide, or to become embryos but never implant. Arthur Caplan, a bioethicist at the University of Pennsylvania, considers it central to the stem-cell debate that "an embryo cannot in itself be considered a human life because as many as half of embryos never become babies." Perhaps it's true that the soul is bestowed at the instant of conception. But, if so, the divine is pitiless, denying a glimpse of the world to at least half of the souls so conceived. "It just can't be that every fertilized egg is a human life," one mother of three says. "Too many things go wrong for that."
Lee Silver, a Princeton University biologist, argues in the 1997 book Remaking Eden that what happens in the womb is a continuum, lacking any clear jump from cell cluster to sanctity: "There are no isolated moments along the way where you can point at an embryo or fetus and say that it is substantially different from the way it was a few minutes or even hours earlier." Silver notes that the fertilized egg can be frozen, apparently indefinitely, and asks in what sense a handful of cells motionless in liquid nitrogen can be considered alive, even if, for the sake of argument, the soul can be frozen, too.
Because the moral status of the early embryo is, at best, ambiguous, it seems likely society will decide that the first claim lies with those already alive, and permit further experimentation to generate stem cells. "We should respect every embryo," the bioethicist Caplan says, "but I'm not going to look at a person in a wheelchair and say, `Sorry, you have to stay in that wheelchair for the rest of your life because of my belief that the frozen embryos in my liquid nitrogen might have become life.'" Thomson contends that stem-cell research is simply inevitable: "Even if we force it out of the United States and the European Union, it will still happen." Stem-cell medicine is coming, ready or not.
There is one possible avenue of escape from the moral enigmas of this research: if stem cells can be found in adults, there will be no need to draw on embryonic "resources." Biology textbooks call this quest hopeless, since by adulthood every cell is differentiated and incapable of further transformation. At least that was the view until February 1997, when the British researcher Ian Wilmut cloned Dolly. Essentially, he did this by taking cells from an adult lamb, making them act like stem cells, and then fusing their DNA into a donor egg which germinated into a baby lamb genetically identical to the adult. Mainstream biologists had thought that embryos, still rich in stem cells, might be cloned, but never adults--or that, if adult stem cells could be found, they would be incapable of reactivating. Since Dolly, however, cows and mice have been cloned from adult cells by variations on the Wilmut technique. Researchers are now finding indications that small amounts of stem cells continue to exist, overlooked, in the adult's nerve tissue and elsewhere; it may be that there are small adult stem-cell deposits throughout the body.
One biotech company, Advanced Cell Technology, claims it has already grown human stem cells by starting with adult tissue. The company says it removed the DNA from a skin cell of a man, inserted these genes into a cow's egg cell in such a way that the human DNA took over, and then watched as the cow egg dutifully produced human stem cells. Word of this procedure, mingling human and animal reproductive cells, caused President Clinton to say he was "deeply troubled" by the experiment-- though, if the work was done as the company claims, the tissues created were human, since the human DNA had taken over.
Several experts dispute whether the experiment actually happened, and Advanced Cell Technology hasn't yet published the peer-reviewed data that back up a discovery claim. But whether or not stem cells have been made from an adult, some biologists are beginning to think this will happen eventually--"eventually," at the current pace of biotech advances, often meaning "next year." Stem cells derived from adults would not only resolve qualms about embryonic tissue; they might have superior therapeutic properties. Suppose you had a liver disease. If one of your own cells could be used as the template for fresh stem cells that would then be converted into liver tissue, what would end up transplanted into you would contain your own DNA and antigens, which presumably would forestall tissue rejection. Transplants might become something the typical person experienced several times in adulthood. Life expectancy would shoot upward, along with the health care share of GDP.
If any person's cells can be made into stem cells, gene therapy and gene engineering, hyped but so far very rare, might also become more common. For two decades, researchers such as W. French Anderson of the University of Southern California have labored to use genes to cure fatal childhood genetic conditions. It just hasn't worked. So far, the only way to deliver healthy DNA into patients is by using viral envelopes as little biological guided missiles. But the immune system shoots these missiles down, defeating the therapy. Anderson, a sainted physician who has spent most of his professional life seemingly inches shy of a great breakthrough, and who instead endlessly watched young patients die, has grown so frustrated he recently proposed attempting gene therapy within the womb. "Once the child is older, the cells may just be too developed to accept new genes," Anderson says--"older" in this sense meaning anyone who's been born.
In 1995, an NIH study panel called genetic therapy "a logical and natural progression in the application of fundamental biomedical science," which is certainly an engaging use of the term "natural." But there's no reason in principle to fear genetic theory, which simply represents using brainpower against medical problems that either God or evolution left for us to solve. And, if stem-cell research succeeds, the barriers against gene therapy may fall. Stem-cell based genetic therapy agents would contain the patient's own DNA code, with only the defective gene altered; the delivery vector would be not viruses but stem cells, which are designed to multiply like mad anyway. At the current rate of progress in research, stem-cell based gene therapy may not be far off, bringing cures to people now classified as incurable.
And once you're standing on this hill, it's pretty easy to see over to the next peak: genetic engineering. Technically, genetic therapy would be a form of genetic engineering--the purpose would be to engineer out the recessive gene that causes cystic fibrosis or sickle-cell anemia, replacing it with healthy DNA that does not. Yet the same technology that may make this gene manipulation possible might make possible the substitution of new genes for DNA that isn't diseased. "DNA engineering itself is now straightforward," says Austin Smith, a biologist at the University of Edinburgh, though only for plants and mice. Plant breeders have been recombining DNA for a decade now, famously inserting fish genes into tomatoes so that they resist frost better, while many of the specialized types of mice used in lab experiments have engineered traits.
Though no gene engineering has been attempted on humans, there appear to be no special technical barriers against doing so. Silver, the Princeton biologist, argues that our generation will be looked back on as "the point in history when human beings gained the power to seize control of their own evolutionary destiny." Control over evolution might turn out well or badly, but there's no reason to reject it out of hand, since evolution has left humanity disease-prone, short-lived, and fragile. Why shouldn't we try to change that?
The disturbing question is whether we will begin sooner than we think through the consequences. Currently, NIH protocols--but not legislation--ban any attempt to alter the human "germline," meaning to change genes in a way that would be passed along to offspring. Gene therapy attempts have been confined to "somatic" cells, the nonreproductive parts of the body, with anything else considered taboo. Yet, last year, Louise Markert of the Duke University Medical Center asked the Recombinant DNA Advisory Committee, which supervises genetic experiments, to begin a debate on whether engineering of reproductive cells should be tested. It's not even the twenty-first century yet, and already earnest, respectable researchers are talking about altering human heredity.
Now back to that cow's egg experiment that may or may not have succeeded. Chromosomes from a grown person were fused into a into a donor egg, the egg began to germinate, and then stem cells were extracted. Suppose the stem cells hadn't been extracted. What might have developed might have been the embryo of a human clone.
Promising as stem-cell research is, what it's doing in the larger scheme is accumulating the technical information that will make human cloning possible. In principle, stem-cell technology might allow a clinic to isolate an adult's gene endowment, engineer it for frost resistance or God knows what else, and then, by allowing the new stem cells to reproduce themselves before implantation, clone unlimited facsimiles of the person.
It may be that the knowledge of cloning is unstoppable, in the sense that no force has ever incarcerated knowledge. And cloning should not be feared in and of itself, for there are arguments in its favor. Clones would be facsimiles of their parents (technically, parent) only in the physical sense. They'd be born as babies--no imaginable technology would create life directly as adults, making the business about rich men or dictators Xeroxing themselves a Hollywood silliness that detracts from the serious arguments against cloning. Character- shaping effects of each generation's particular upbringing would inevitably make the cloned child differ from the parent, while a clone's thoughts, personality, and experiences would reflect the unique human dignity possessed by every individual. No one argues that each member of a pair of identical twins, who are genetic duplicates, does not possess unique dignity.
Human cloning would also have the beneficial effect of providing the means for even the infertile person to conceive and raise a child, a blessing it is easy for the fertile to overlook. Thus we should not be squeamish about cloning merely because it mixes reproduction, technology, and the new. Leon R. Kass has argued in these pages (see "The Wisdom of Repugnance," tnr, June 2, 1997) that cloning is horrifying in part because it would make possible "the grotesqueness of conceiving a child as an exact replacement for another who has died." Aside from the fact that child B would not and could not be "an exact replacement," as both a parent and a churchgoer this possibility does not strike me as grotesque. If a child's life were cut short, the prospect that the lost child could leave behind a similar life, consoling the parents and preserving the memory of child A, might seem like a miracle.
When IVF techniques made their debut in 1979, pundits pronounced themselves repelled by "test tube" children, and polls showed the public strongly opposed. By 1994, at least 150,000 IVF babies had been born, brightening the lives of couples who would otherwise have been barren, while public support for IVF science had become widespread. Babies made possible through this technology aren't weird or abnormal, and they are less likely to suffer from gene-defect diseases, since their embryonic chromosomes are comparatively easy to check. The first American IVF child, Elizabeth Carr, just turned 17, and she wants to be a journalist. According to The Boston Globe, she attends an annual reunion of IVF children and delights in holding the latest in her lap. Can anyone believe the world would be a better place if the technology that caused Elizabeth Carr had been banned?
Every new life has an interest in being born, which was the clincher argument for IVF, and may someday be the clincher argument for cloning. Imagine meeting a cloned person and asking, "If you had been given the choice of either not existing or being conceived as a clone, which would you have chosen?"
Of course, it's possible the cloned person would turn the question around and say, "If you had been given the choice of either coming into existence as an unpredictable one of a kind or as a clone, which would you have chosen?" Perhaps there are some people with genetic defects who would say they'd rather have been cloned from someone with problem- free DNA, but most of us would say we'd rather have our gloriously unpremeditated forms. Cloning should produce physically healthy children--but what about mentally healthy ones? It's one thing to bear resemblance to a mother or father; most children take pride in that. It may be another to be born with expectations about you preformed--to miss the chance, as Kass has beautifully said, to arrive "an unbidden surprise, a gift to the world."
If a child could only be conceived through cloning because circumstances made it impossible for the parent to have progeny any other way, then the clone's interest in being born might outweigh the psychological risks. But what about the prospective mother or father who simply wants a mirror-image child, especially if the motive is vanity? In that case the chance of harmful psychological burdens on the child might argue against allowing cloning. It is this kind of imaginable-now issue, rather than science fiction about biotech dystopias, that ought to govern public debate on stem cells and the prospect of cloning.
Today's situation with stem cells and cloning might be likened to what would happen if a fleet of modern jet fighters were teleported back in time to ancient Sumeria. First, the ancients would marvel at the objects, noting their extraordinary complexity--as scientists marveled when they first glimpsed the extent of the double helix. Initially, they'd be too scared to touch, and some would argue that the gods would punish those who touched. Eventually, the fear would wane, and, by poking and prodding and pushing buttons, someone would manage to start one of the plane's engines, generating thunder and fire. At that point, the ancients would believe they had "discovered" the true purpose of the mysterious objects, and that, now being able to manipulate the planes, they had become masters of them.
Owing to the stem-cell breakthrough, there now stands the prospect that our children will not only live healthier lives but that their children will be the final generation of Homo sapiens, supplanted by Homo geneticus or whatever comes next. Homo erectus didn't last, so there's no reason to assume Homo sapiens won't ever give way to a next stage. If all goes well, the advent of control over our own cells might offer our grandchildren many things we would wish for them.
But it's all happening much, much faster than society understands. It's also happening under conditions in which we are telling ourselves that we understand genes because we have learned to make them do certain things, but we probably know little more about the totality of our DNA than would the ancient who doesn't even realize that airplanes are supposed to fly. It's time to move biotechnology to the center of the national debate, so that we can sort out its rights and wrongs before sheer technological momentum imposes an outcome upon us.
(Copyright 1999, The New Republic)
FROM: http://www.greatdreams.com/lcfrclnng.htm
TEST TUBE BABIES
In vitro fertilisation (IVF) is a process by which an egg is fertilised by sperm outside the body: in vitro. IVF is a major treatment for infertility when other methods of assisted reproductive technology have failed. The process involves monitoring a woman's ovulatory process, removing ovum or ova (egg or eggs) from the woman's ovaries and letting sperm fertilise them in a fluid medium in a laboratory. When a woman's natural cycle is monitored to collect a naturally selected ovum (egg) for fertilisation, it is known as natural cycle IVF. The fertilised egg (zygote) is then transferred to the patient's uterus with the intention of establishing a successful pregnancy. The first successful birth of a "test tube baby", Louise Brown, occurred in 1978. Louise Brown was born as a result of natural cycle IVF. Robert G. Edwards, the physiologist who developed the treatment, was awarded the Nobel Prize in Physiology or Medicine in 2010.
The term in vitro, from the Latin meaning in glass, is used, because early biological experiments involving cultivation of tissues outside the living organism from which they came, were carried out in glass containers such as beakers, test tubes, or petri dishes. Today, the term in vitro is used to refer to any biological procedure that is performed outside the organism it would normally be occurring in, to distinguish it from an in vivo procedure, where the tissue remains inside the living organism within which it is normally found. A colloquial term for babies conceived as the result of IVF, "test tube babies", refers to the tube-shaped containers of glass or plastic resin, called test tubes, that are commonly used in chemistry labs and biology labs. However, in vitro fertilisation is usually performed in the shallower containers called Petri dishes. One IVF method, autologous endometrial coculture, is actually performed on organic material, but is still considered in vitro.